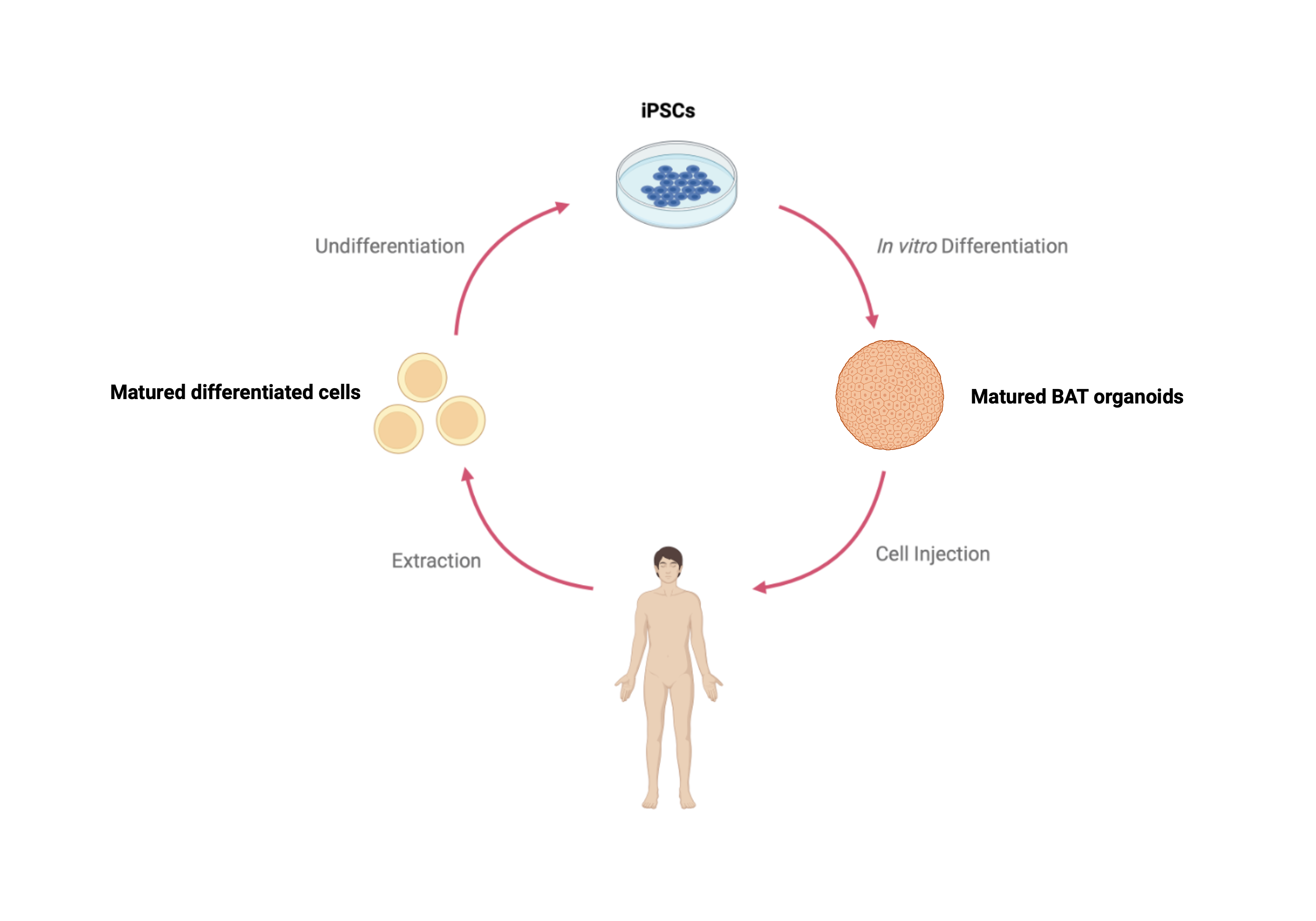
Brown adipose tissue (BAT) plays a crucial role in energy regulation and adaptive thermogenesis generating heat through UCP1 activation. This unique capability makes BAT a promising therapeutic target for metabolic diseases such as type 2 diabetes, obesity, and other related metabolic disorders. Understanding the developmental pathways of BAT is critical for designing novel therapeutic strategies aimed at enhancing its regenerative potential or functional activity. Recent advances in in vitro differentiation protocols, such as the approach developed by Jyoti Rao, have enabled the generation of brown adipocytes from induced pluripotent stem cells (iPSCs) by imitating key steps of BAT developmental pathways. However, significant limitations remain. These include the prolonged duration required for differentiation and the challenges of engrafting these adipocytes due to their fragile structure as single cells. Consequently, traditional cell therapy approaches, which rely on dissociating cells into single-cell suspensions for injection, are not feasible for effective BAT transplantation. To address these limitations, we propose using the “somitoids” model developed by Yuchuan Miao. Somitoids are three-dimensional structures that recapitulate early embryonic somite formation, a critical stage in the derivation of BAT precursors. By using somitoids as a starting point, we aim to reconstruct the full developmental pathway leading to the formation of BAT organoids. This novel approach has the potential to produce more physiologically relevant BAT tissue, both structurally and functionally, compared to current methods.
December 28, 2025