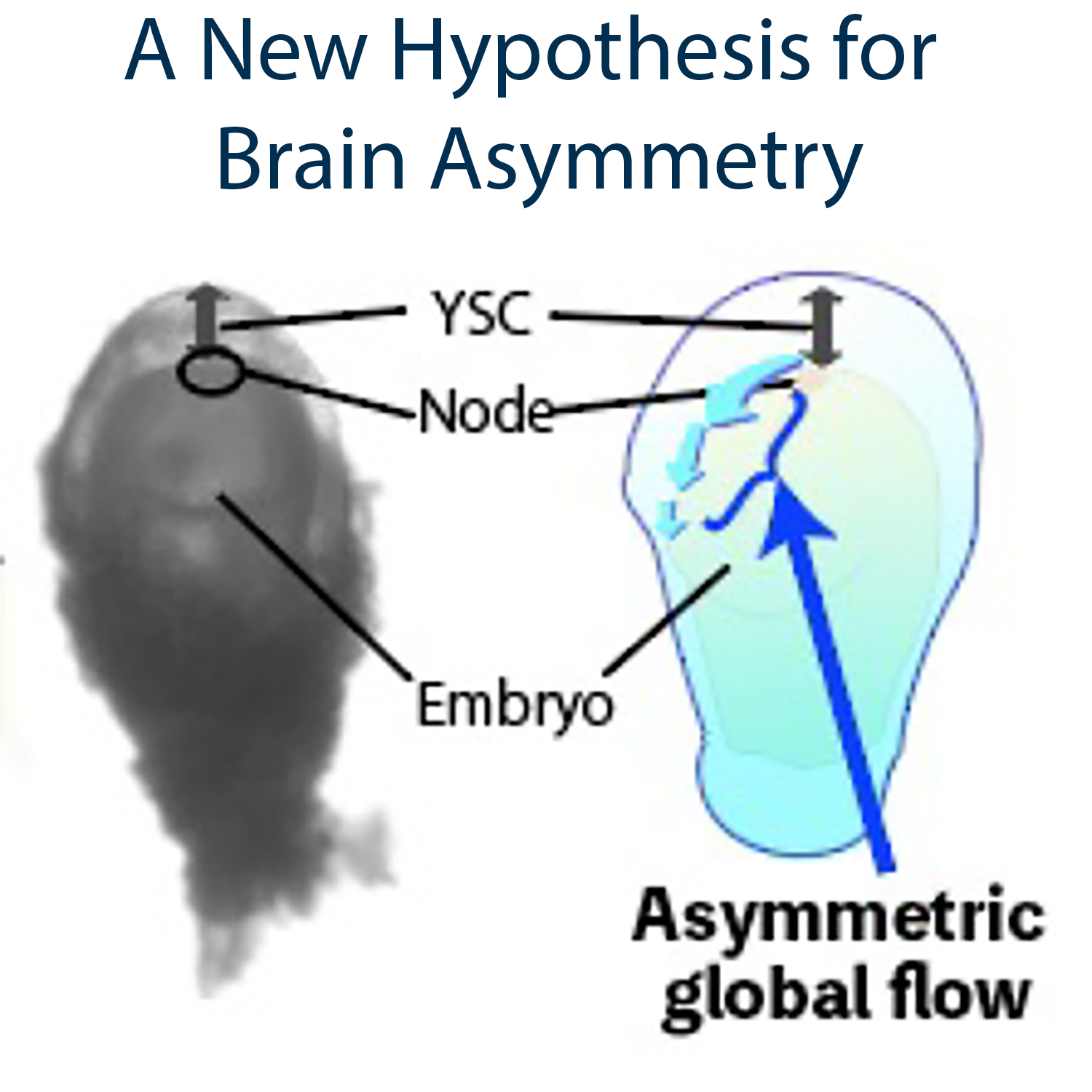
The human brain exhibits striking left-right asymmetry in both its functions and anatomy, but the mechanisms underlying this asymmetry during embryonic development remain poorly understood. Here, we propose a novel hypothesis, combining computational fluid dynamics with mouse developmental biology, suggesting that directional fluid flow around the early embryo may contribute to the establishment of left-right asymmetry in the brain. In contrast to the brain, the mechanisms underlying left-right asymmetry in the visceral organs, such as the heart and liver, are well-established. In contrast to the brain, visceral organs such as the heart and lungs exhibit clear left-right asymmetry. In mammals, the first symmetry-breaking event occurs in the embryonic node, where ciliary rotation drives leftward fluid flow. This flow generates subtle asymmetries in force distribution near the node, which are subsequently amplified and integrated into downstream gene regulatory networks. Although this left-right symmetry breaking is typically considered independent of brain asymmetry, little is known about its potential influence on the brain. Through our investigation in mouse embryos, we discovered that the node is connected to the large extraembryonic space, the yolk sac cavity. This observation led us to hypothesize that cilia-driven flow at the node could extend beyond the node, inducing global fluid movement that may contribute to the establishment of left-right asymmetry in non-visceral organs, including the brain. Given the challenges of directly measuring fluid dynamics within the yolk sac cavity, we employed computational fluid dynamics to simulate cilia-driven flow. We examined how the spatial patterns of ciliary rotation influence fluid dynamics and whether such directional flow could propagate across the entire embryo. Our simulations suggest that nodal cilia can indeed induce a global flow, potentially contributing to LR asymmetry in the brain. This presentation will address our ongoing efforts to validate this hypothesis through in vivo measurements of fluid dynamics.
December 28, 2025