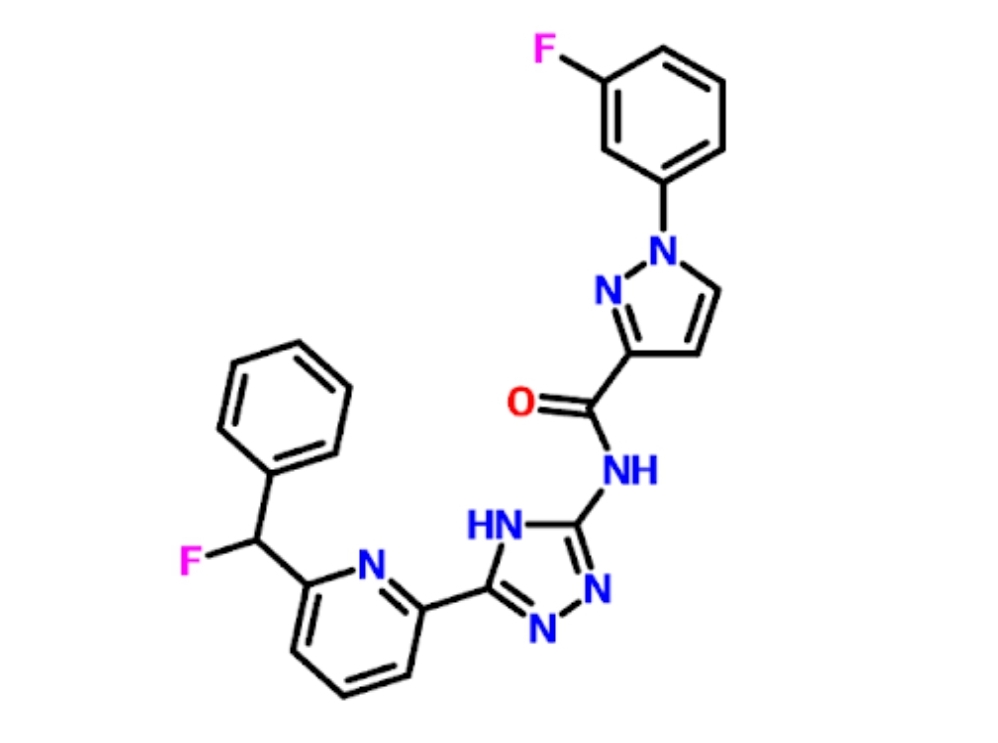
Single mutations in the SOD1 protein, such as A4V and D90A, are known to induce aggregation of Superoxide dismutase 1 (SOD1) proteins, which is often universal for an amyotrophic lateral sclerosis (ALS) diagnosis. The D90A mutation is the most common of the ALS-causing point mutations and is known to show significant aggregation potential. This in silico inquiry was conducted on the D90A variant of the SOD1 protein, to create a molecule that could potentially inhibit aggregation. Through qualitative examination of the structures, it was determined that the residue Tryptophan 32 is an ideal target for the proposed small molecule. It differs sterically from the D90A mutant to the wild-type and is also distant from the dimerization and allosteric sites. These properties make it unlikely that the small molecule will interfere with the enzyme’s regular function when bound. No-Mo-Aggregate-81 (NMA81), a novel small molecule that specifically targets the Trp 32 residue, is proposed as potential treatment to prevent aggregation of the SOD1 proteins. Reduced aggregation of SOD1 is expected to be associated with decreased severity of ALS.
December 28, 2025