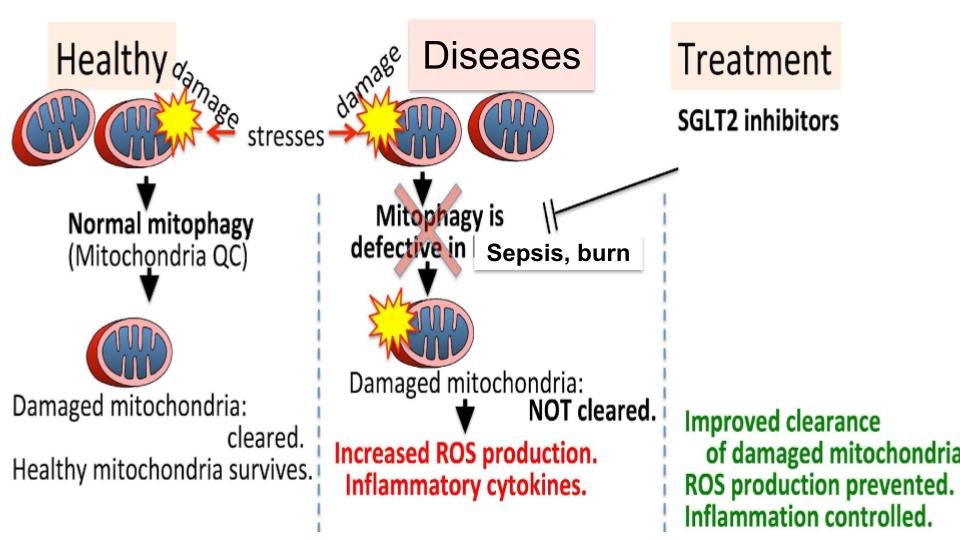
Mitophagy, the selective autophagic degradation of mitochondria, is a conserved quality control mechanism essential for cellular and mitochondrial homeostasis. Its dysfunction contributes to various human diseases, but the precise role of microtubule (MT) network dynamics in regulating mitophagy under stress remains poorly understood. Inspired by Lewis Carroll’s concept of the Red Queen’s race, which symbolizes maintaining equilibrium under constant demand, this study examines how cytoskeletal dynamics influence mitophagy across two distinct stress platforms: burn serum and lipopolysaccharide (LPS)-induced conditions.
In this cross-platform investigation, we utilized C2C12 myocyte cell lines expressing mRuby-tubulin, GFP-EB1 (a marker of MT plus ends), and mito-Keima or mitoKaede. These tools enabled live-cell monitoring of MT synthesis speed, network formation, and mitophagy flux. Stress models were created using rat-derived burn serum (30% body surface area) and LPS (1 μg/mL) to mimic critical illness conditions. MT synthesis rates were tracked via EB1 motion, basal and stress-induced MT network formation were analyzed, and mitophagosome maturation was assessed through ratiometric imaging of mitophagy reporters.
Under both burn serum and LPS-induced stress conditions, MT synthesis rates decreased significantly compared to controls. Basal MT network formation showed no differences between stressed and control groups; however, MT deficiencies became evident following CCCP-induced mitophagy. Mito-Keima and mitoKaede analyses revealed impaired vesicle trafficking, characterized by defective autophagosome/mitophagosome/lysosome interactions and diminished mitophagosome maturation. These findings indicate that MT network abnormalities manifest primarily under conditions of increased mitophagy flux demand, where robust cytoskeletal dynamics are critical for maintaining functionality. This study introduces the concept of the “Red Queen’s race” as a framework for understanding the interplay between cytoskeletal dynamics and mitophagy regulation. The analogy underscores the need for continuous, vigorous MT synthesis to sustain equilibrium, particularly under stress. The observed cytoskeletal dysfunction in both burn and LPS stress models highlights a shared mechanism linking stress-induced MT defects to inadequate mitophagy responses. By bridging these two distinct platforms, this work reveals that stress-induced cytoskeletal defects disrupt mitophagy flux through impaired vesicle trafficking and maturation, offering a unifying perspective on the impact of MT network dysfunction in critical illness. These insights provide a novel understanding of how cytoskeletal dynamics govern mitochondrial quality control, emphasizing the potential for targeting mitophagy maturation as a therapeutic strategy across diverse pathological conditions.
December 28, 2025