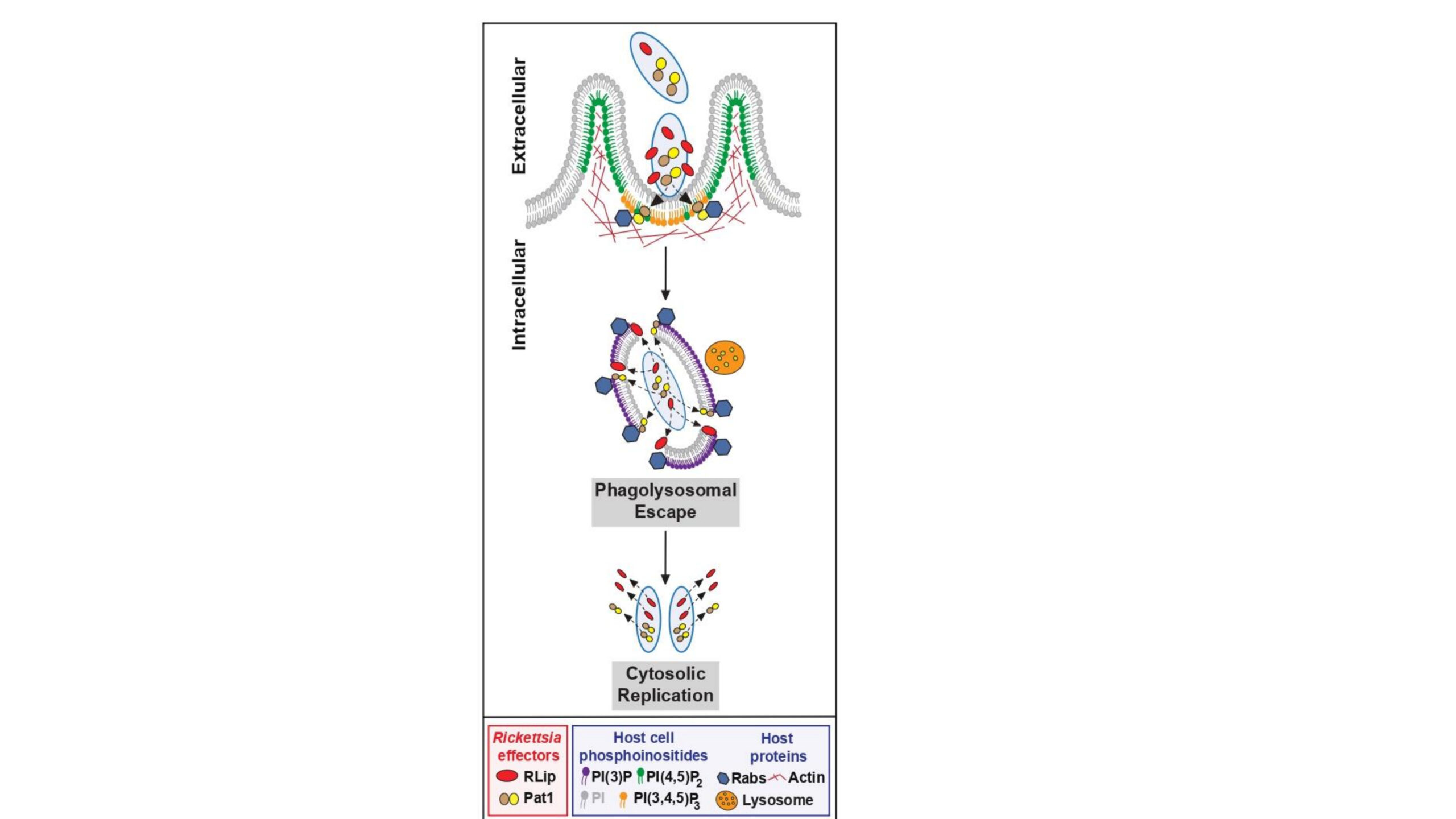
Key cellular processes for the rickettsial obligate intracellular lifestyle, include internalization by phagocytosis, regulation of intracellular trafficking, and evasion of lysosomal destruction to establish an intracytosolic replication niche, remain poorly defined. Recent reports showed that rickettsial phospholipases play an important role in vacuolar escape, but their functions are dispensable depending on the host cell-type. Here, we report the identification of a putative lipase with a Serine hydrolase motif (GXSXG) in the R. rickettsii genome, which we named RLip (Rickettsia Lipase). Sequence comparison shows that the Serine hydrolase motif is conserved among RLip molecules of other Rickettsia species. Our work reveals that RLip harbors a lipase activity, and its recombinant expression is cytotoxic to yeast and mammalian cells. We further demonstrate that RLip expression is induced during R. rickettsii or R. parkeri infection, while its expression was minimally detected during R. montanensis (non-pathogenic) infection. Fractionation of R. rickettsii-infected host cells, shows the presence of RLip in the cytoplasmic fraction, while being minimally retained by the bacteria. Infection studies in HMEC-1 cells using R. parkeri wild type (WT) or R. parkeri rlip::Tn (non-functional RLip), demonstrate that lack of RLip function significantly impairs rickettsial evasion from bactericidal phagolysosomal fusion, suggesting that RLip plays a critical role in the escape from membrane-bound vacuoles to facilitate the intracytosolic colonization of pathogenic Rickettsia species
Keywords: Phagocytosis, Rickettsia Lipase, Pathogenic