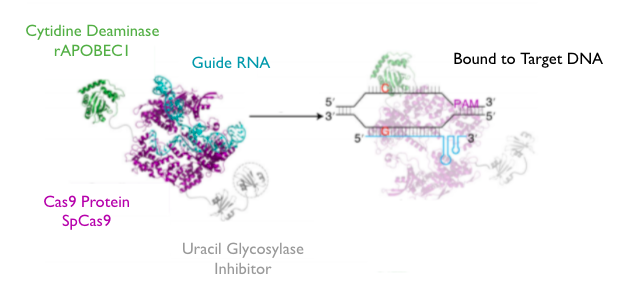
Gaucher disease is a genetic disorder caused by point mutations in the GBA gene encoding for glucocerebrosidase, an enzyme that breaks down the fat glucocerebroside. This work proposes the development of a Cas9-cytidine deaminase fusion protein to treat forms of Gaucher disease type 1 associated with an A1226G substitution on the GBA gene. This fusion protein would consist of the rAPOBEC1 cytidine deaminase, the Streptococcus pyogenes Cas9 nickase, and a uracil glycosylase inhibitor (UGI) connected with flexible serine/glycine linkers. This fusion protein, once complexed with an engineered guide RNA, would specifically bind to the mutated site near the Protospacer Adjacent Motif (PAM) and enzymatically convert the disease-associated cytosine base to thymine, reversing the disease mutation. The proposed system is unlikely to exhibit unintended edits; the designed guide RNA does not align with any other known locus on the human genome. Ultimately, we imagine expressing the fusion protein/guide RNA complex in vitro, encapsulating it in lipid nanoparticles, and injecting it into the body; to reach this goal, we propose three specific experimental aims: (1) expressing and purifying the fusion protein and guide RNA; (2) demonstrating consistent and on-target editing activity in human macrophage cell cultures; and (3) demonstrating successful delivery and on-target editing activity in a model organism. The completion of this project would put forth a vastly more accessible treatment for Gaucher disease type 1 and advance the precision of Cas9-based gene editing.
December 28, 2025