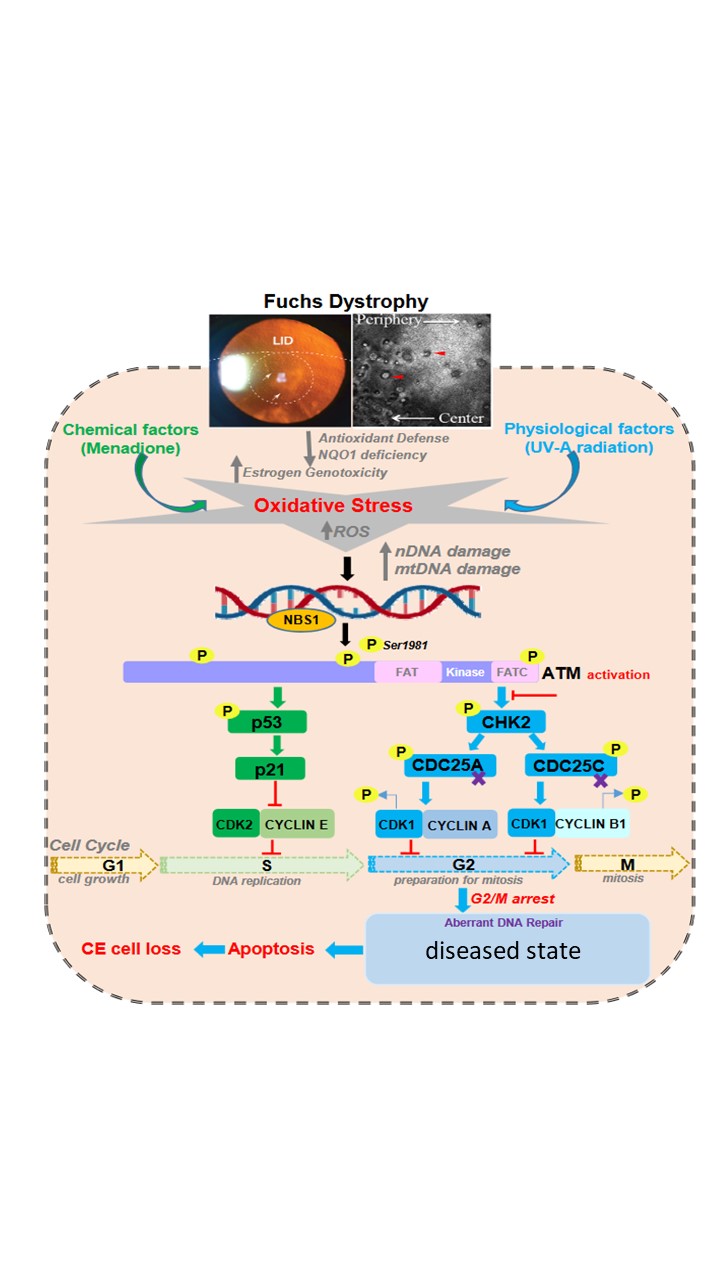
Purpose: Fuchs Endothelial Corneal Dystrophy (FECD) is an oxidative stress disorder leading to accelerated loss of corneal endothelial cells (CEnCs) and exhibits greater accumulation of DNA damage compared to normal aging CEnCs. Ataxia Telangiectasia Mutated (ATM) is a key DNA damage response (DDR) kinase in post-mitotically arrested cells, which is activated and recruited to the damage site by Mre11-Nbs1-Rad50 (MRN) complex, leading to downstream activation of cell cycle checkpoints, DNA repair and/or apoptosis. In this study, we investigate the mechanism of ATM-mediated DDR activation in response to UVA leading to FECD.
Methods: To investigate DDR activation in response to UV irradiation in vitro, immortalized normal and FECD patient-derived cell lines were treated with UVA (10 J/cm2) + 4-OHE2 followed by recovery for 1 and 24 hours in serum-free media. Cells were lysed at 1-hr post-treatment to compare early DDR signaling between cell-lines. The phosphorylation status and levels of γH2AX (Ser139) and pATM (Ser1981) and its downstream effectors pCHK2 (Thr68), caspase-3 and caspase-9 were evaluated by western blotting. Cell-cycle distribution was measured 24-hrs post-treatment by flow cytometry. The effect of cell cycle reentry on DNA repair efficiency was determined by sorting the treated cells into G0/G1-G2/M with MoFLo and alterations in different DNA repair genes were quantified by qPCR. In vivo, cell cycle activation, DNA damage and senescence were tested by Ki67, H3K9Me2 and TUNEL staining in CEnCs using whole mount corneas (+/- UVA; 500 J/cm2) of wild-type (WT) and ATM knockout mice.
Results: We detected increased activation of pATM/ATM, an early responder to DNA damage, and its effector pCHK2, γH2AX, caspase-3 and caspase-9 in FECD compared to normal CE cells after treatment with UVA + 4-OHE2, indicating that ATM possibly activates pCHK2 preferentially by direct phosphorylation in FECD. UVA+4-OHE2 treated FECD cells were arrested in G2/M phase of the cell cycle. Gene expression analysis of important DNA repair genes (LIG3, NEIL2, TOP3A, XRCC1 and XPC) in G2/M phase-arrested UVA-treated CEnCs, compared to untreated, showed their significant upregulation, highlighting that UVA-mediated DNA-repair can be induced by holding cells in G2/M, irrespective of DNA damage. In vivo, UVA exposure was found to promote cell cycle reentry post one-day in WT but not ATM-knockout mice. At 1-month post-UVA WT mice showed increased senescence while ATM-knockout mice presented with induction of apoptosis.
Conclusions: We demonstrate increased activation of ATM-mediated DDR in response to ROS-induced DNA damage in FECD. This study provides insights into understanding the role of DDR signaling cascade in determining the apoptotic cell fate of CEnCs in FECD.